PDF - Review
Review
 Document
Document
Modular dual mobility (MDM) hip systems: Is there a risk of fretting corrosion?
 Document
Document
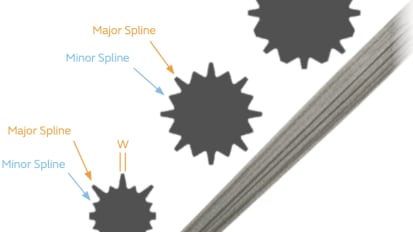
ROCKTITE™ Brochure: Is Your Fixation ROCKTITE™?
REDAPT™ is a 3° tapered titanium stem with proprietary ROCKTITE™ distal flutes designed for stable diaphyseal fixation through major and minor splines.
 Document
Document
REDAPT™ Revision Femoral System Demonstrates Stable Fixation
This is a multicentre, retrospective study investigating short-term outcomes in 153 consecutive patients receiving REDAPT™ Revision Femoral System.
 Document
Document
REDAPT™ Revision Femoral System Demonstrates Favourable Outcomes
The READAPT™ Femoral Revision System demonstrates favourable early clinical, radiological and patient-reported outcomes in revision total hip arthroplasty (rTHA).
 Document
Document
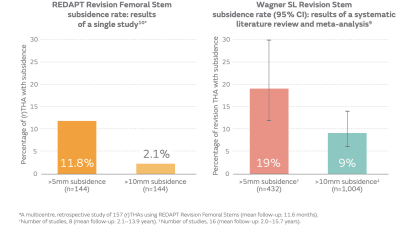
REDAPT™ Revision Femoral System Decreases the Incidence of Subsidence
This retrospective, observational study compares rates of subsidence >5mm in modular versus non-modular tapered, fluted, titanium (TFT) stems.
 Document
Document
REDAPT™ Revision Femoral Stem Subsidence Evidence
READAPT™ Revision Femoral Stem -- stable fixation with low subsidence at 1 year.
 Document
Document
REDAPT™ Revision Femoral Monolithic Surgical Technique
The technique description herein is made available to the healthcare professional to illustrate the suggested treatment for the uncomplicated procedure. In the final analysis, the preferred treatment is that which addresses the needs of the patient.
 Document
Document
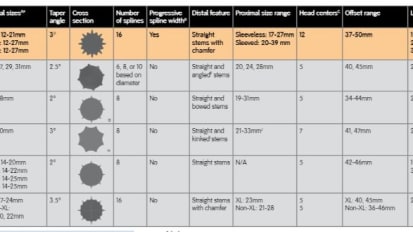
REDAPT™ Revision Femoral Competitive Matrix
This chart is a competitive matrix in relationship to Smith + Nephew's READAPT™ Revision Femoral System.
 Document
Document
REDAPT™ Revision Acetabular Stiffness Comparison
This document explores the stiffness comparison of REDAPT™ Locking Screws and non-locking screws utilized with the REDAPT™ variable angle locking feature.
 Document
Document
REDAPT™ Revision Acetabular Shell Surgical Technique
This document highlights the surgical technique when using the Redapt™ Revision Acetabular Shell.
 Document
Document
REDAPT™ Revision Acetabular Design Rationale
The revision shells are developed for use in revision cases where compromised bone makes implant fixation and stability more difficult.
 Document
Document
REDAPT™ Revision Acetabular Augment Design Rationale
The REDAPT™ Acetabular Augments are developed for use in revision total hip arthroplasty cases where bone voids exist that may not be able to be addressed solely through placement of an acetabular shell.
 Document
Document
REDAPT™ Modular Shell Surgical Technique
The technique description herein is made available to the healthcare professional to illustrate the manufacturer’s suggested treatment for the uncomplicated procedure.
 Document
Document
REDAPT™ Fully Porous Shell with Polarcup
Instability after primary and revision total hip arthroplasty (THA) is a leading cause for revision.
 Document
Document
REDAPT™ CONCELOC Pre-Clinical Animal Study
Osseointegration of the CONCELOC™ Advanced Porous Titanium was assessed in a previously validated, load-bearing ovine model.
 Document
Document
REDAPT™ Blade Augment: Static and Fatigue Strength Testing
Buttress-type augments have successfully been used as part of acetabular revision surgery to prevent shell migration in severe acetabular defects (Paprosky Type IIIA/IIIB) with good short to mid-term outcomes.
 Document
Document
REDAPT™ Acetabular Augment Surgical Technique
This document presents the surgical technique when using the Redapt™ Revision Acetabular System.
 Document
Document
REDAPT™ Acetabular Augment Sizing Wall Chart
This is a helpful Redapt™ Acetabular Augment Sizing Wall Chart.
 Document
Document
A single centre, multi-surgeon, retrospective observational study on patients who underwent primary or revision THA with the uncemented R3™ Acetabular System between January 2009 and December 2012 with a minimum of 7 years follow-up.
 Document
Document
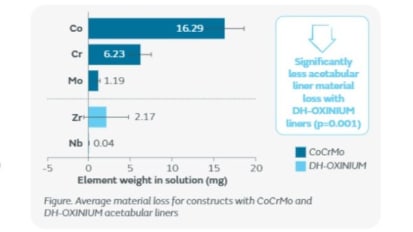
Contructs with DH-OXINIUM liners demonstrated lower material loss than those with CoCrMo liners.
 Document
Document
As the number of cases and the age of patients broadens, orthopaedic applications demand a high performance implant material that can deliver proven clinical performance in hip and knee arthroplasty.
 Document
Document
OXINIUM™ Technology in Total Hip Arthroplasty
Exclusive to Smith+Nephew, OXINIUM™ Technology is an award-winning, advanced implant material available for hip and knee arthroplasty.
 Document
Document
The OR3O™ Dual Mobility System is intended for use in primary and revision total hip arthroplasty in skeletally mature patients.
 Document
Document
REDAPT™ Acetabular is lateralized at the base 4mm. By switching from R3™ to REDAPT™, your surgeon gains 2mm of offset.
 Document
Document
This is an OR3O™ Dual Mobility with OXINIUM™ DH Linear Technology tray layout guide with ordering information.
 Document
Document
Modular Dual Mobility and Corrosion
Dual mobility (DM) acetabular components have been designed to deliver increased range of motion with good stability, whilst reducing wear in primary and revision total hip arthroplasty (THA) compared to standard implant designs.
 Document
Document
Dual mobility (DM) bearings are cost effective in patients aged <75 years undergoing revision total hip arthroplasty (rTHA) over an initial 5 year period, and cost saving from 10 years compared with conventional bearings
 Document
Document
CONCELOC™ Material Specifications
Through our pioneering approach to design, Smith & Nephew engineers have developed a patented method for creating a fully randomized porous structure with predictable porosity, pore size and node interconnectivity.
 Document
Document
Malseated liners may have a higher propensity for micromotion and corrosion leading to increased wear and osteolysis.
 Document
Document
The REDAPT™ Fully Porous Shell is fabricated through additive manufacturing, which permits greater design flexibility compared to standard, subtractive manufacturing (i.e., machining).
 Document
Document
2-Year Retrospective Study of OR3O™
The OR3O™ Dual Mobility System is a new modular system designed for use in patients at risk of instability following THA, available from January 2020 in the US.
 Document
Document
Computer-guided THA is associated with reduced risk of revision and increased patient satisfaction compared to conventional THA in the National Joint Registry (NJR) of England, Wales and Northern Ireland
 Document
Document
RI.HIP Navigation: Collection of Evidence
THA is recognised as a highly successful operation, with a high survivorship at 15 years. The world's largest registry - the National Joint Registry (NJR) of England, Wales and Northern Ireland - reports.
 Document
Document
R3™ Acetabular System with OXINIUM™ Technology: Optimized XLPE Liner Offering Chart
R3™ Optimized XLPE Liners
 Document
Document
There are several reasons why OXINIUM™ Technology provides benefits beyond traditional cobalt chrome (CoCr) hip and knee components.
 Document
Document
Introducing a New Modular Dual Mobility Acetabular Component: OR3O™
This dual mobility (DM) acetabular component is designed to deliver increased range of motion with good stability to address dislocation whilst reducing wear.
 Document
Document
LEGION™ COCNELOC™ Cementless Total Knee System
The LEGION™ Total Knee System has been designed to offer the orthopaedic surgeon solutions to address intraoperative situations. Implant function is directly related to accurate surgical technique.

